The entire pharmaceutical industry is built on various forms of data and has recently invested substantial resources towards collecting and analyzing real-world data (RWD). RWD has traditionally been viewed as a tool to primarily support market access and demonstrate product value. But when such data is used beyond the traditional applications, throughout the product lifecycle and across different functions, its true potential begins to emerge. That is why every pharma company should have a clear real-world data strategy at both global and local levels.
From data to evidence
Real-world evidence (RWE), the evidence generated based on RWD, has great potential to answer to wealth of essential questions that people in the pharma industry are facing every day. However, the data that is collected for non-research purposes across healthcare systems, or even at patients’ homes, is certainly not the easiest to tackle. That is why it is important to select the best data sources and ensure that the research team has strong experience in handling such datasets. When done properly, RWD projects can be run efficiently, even within months, and with reasonable costs.
For getting solid real-world evidence (RWE), correct interpretation of the results is as important as quality of data. It is important to recognize that real-world studies are set up differently than randomized controlled trials (RCT), and they also answer totally different questions. RWD is not intended to replace RCTs but the evidence from both study types are needed to arrive at a complete set of evidence. Some of the key differences between RCTs and RWE are illustrated in Figure 1.
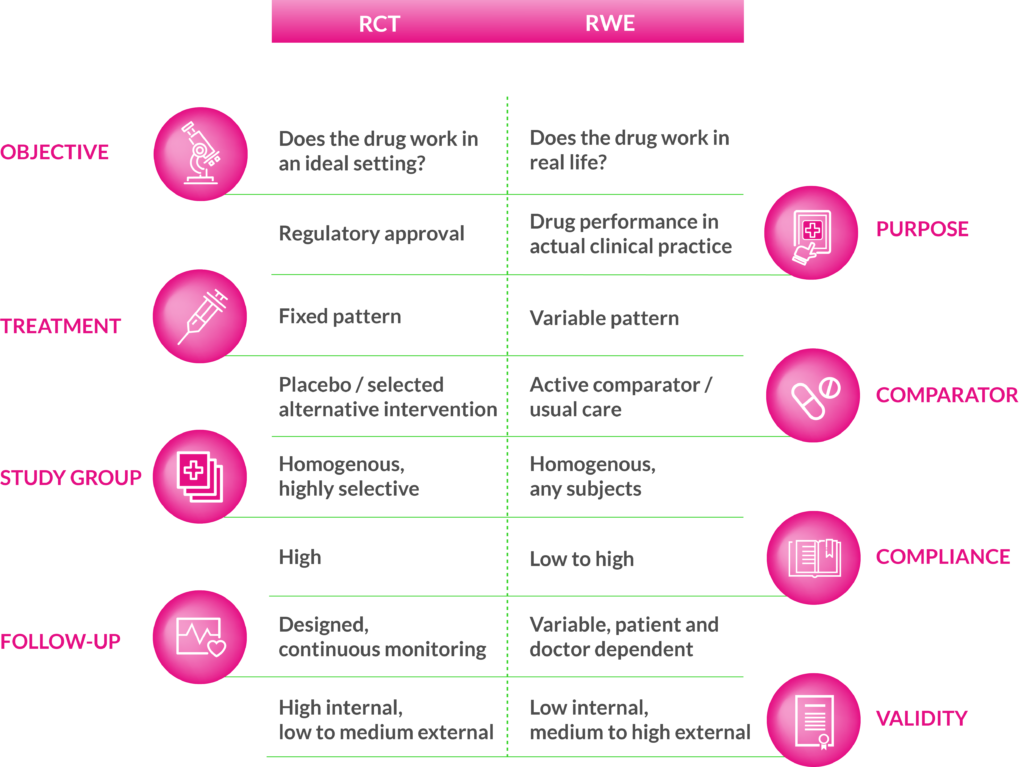
Figure 1. The key differences between randomized controlled trials (RCT) and real-world evidence (RWE)
Capturing the value
By itself, RWD is unlikely to be highly valuable for any company. It is the ability to generate exceptional insights, to implement the data to meaningful activities, and to make better business decisions, which generates the value for an organization. It has been estimated that with broad applications, the value of RWE can be as high as $1 billion annually for each of the largest pharma companies. Implementing a successful RWE strategy will definitely bring significant value also at the local level.
Market access, and to some extent medical affairs, has traditionally been the leader in RWD and continues to have a central role also in the future. But there are endless numbers of other applications for RWE within an organization; some of these are shown in Figure 2.
.
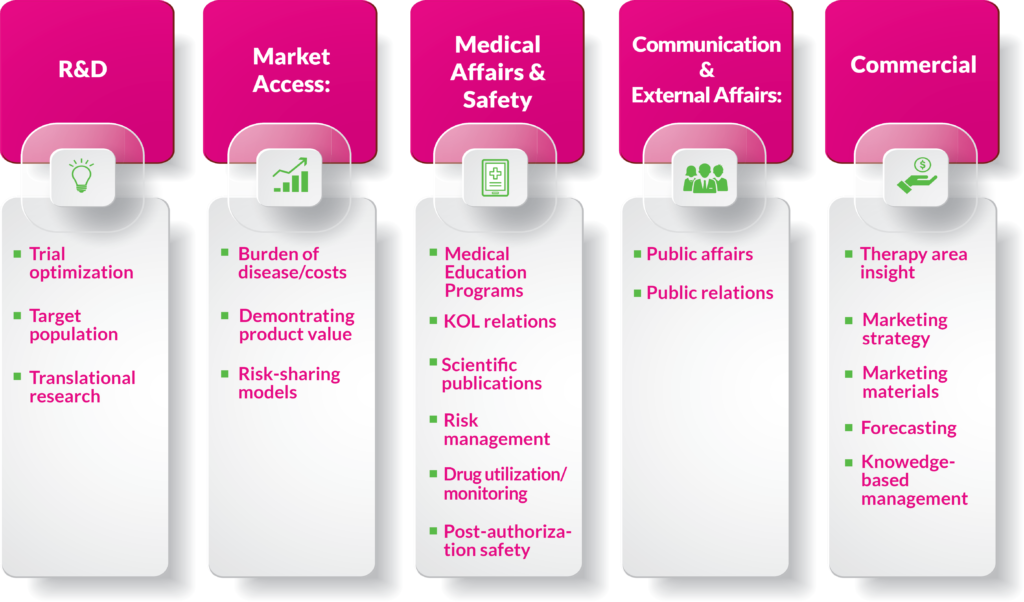 Figure 2. Examples of RWE applications.
Figure 2. Examples of RWE applications.
Building the RWD strategy
Here are some considerations you might want to take into account when designing your RWD strategy:
Be systematic. Take into account the product’s current status, future needs, and the needs of different functions. Set up clear goals, timelines, and approaches. Work on long-term, and avoid ad hoc, projects.
- Design the data implementation. How will the data be converted into action? What are the key insights that you are looking for? These, and many other questions, need to be asked in the planning phase, not when the data is already generated. Think beyond traditional approaches to ensure maximum value of the data.
- Set up the right team. Ensure that your team really knows where to get the right data and how to analyze and interpret it. The team should also have a strategic understanding of data implementation.
- Learn by doing. There is enough talk of the promise of RWD, but you only learn by doing. First-hand experience is the only way to truly understand the value of the data for your organization.
The companies with the best data strategies are likely to be the leaders of the industry of tomorrow. Now is the time to move forward.




